A) N2
B) O2
C) F2
D) all of these
E) none of these
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following molecules has polar bonds but is a nonpolar molecule PCl5, PCl3, NCl3, CO2
A) NCl3 and CO2
B) PCl5 and CO2
C) PCl3 and CO2
D) PCl5, PCl3, NCl3 and CO2
E) None of the above
G) B) and E)
Correct Answer

verified
Correct Answer
verified
True/False
The hybridization of B in the BF3 molecule is sp3.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
N,N-diethyl-m-tolumide (DEET) is the active ingredient in many mosquito repellents. What is the hybridization state of carbon indicated by the arrow in the structure of DEET shown below 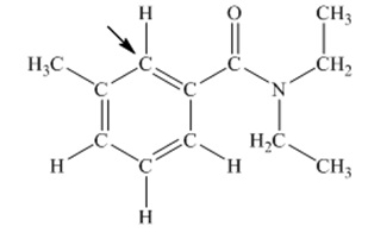
A) sp
B) sp2
C) sp3
D) sp3d
E) sp3d2
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Indicate the type of hybrid orbitals used by the central atom in BrF3.
A) sp
B) sp2
C) sp3
D) sp3d
E) sp3d2
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The hybridization of the central nitrogen atom in the molecule N2O is
A) sp.
B) sp2.
C) sp3.
D) sp3d.
E) sp3d2.
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The number of pi bonds in the oxalate ion (C2O42-) is
A) 1
B) 2
C) 3
D) 4
E) 5
G) B) and C)
Correct Answer

verified
Correct Answer
verified
True/False
In VSEPR theory the molecular geometry of SF4 (sulfur tetrafluoride) is distorted tetrahedron (seesaw).
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The geometry of the hybrid orbitals about a central atom with sp3d hybridization is:
A) linear
B) trigonal planar
C) tetrahedral
D) bent
E) trigonal bipyramidal
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A central atom with 4 electron pairs (single bonds and/or lone pairs of electrons) could have which of the following molecular geometries I. Trigonal bipyramidal II. Tetrahed ral III. Trigonal pyramidal IV. Bent
A) I and II
B) II and III
C) II, III, and IV
D) I and IV
E) I, II, and IV
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Indicate the type of hybrid orbitals used by the central atom in PCl3.
A) sp
B) sp2
C) sp3
D) sp3d
E) sp3d2
G) B) and D)
Correct Answer

verified
Correct Answer
verified
True/False
The bond in O2 is shorter than the bond in O2+.
B) False
Correct Answer

verified
Correct Answer
verified
True/False
The geometry of the hybrid orbitals of the central atom of a molecule is always the same as molecular geometry.
B) False
Correct Answer

verified
Correct Answer
verified
True/False
More energy is required to break a bond with an order of 3/2 than is required to break a bond of order 2.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A central atom with 5 electron pairs (single bonds and/or lone pairs of electrons) could have which of the following molecular geometries I. Trigonal bipyramidal II. Seesaw III. T-shaped IV. Linear
A) I, II, and III
B) II, III, and IV
C) I, III, and IV
D) I, II, and IV
E) I, II, III, and IV
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Give the number of lone pairs around the central atom and the molecular geometry of SCl2.
A) 0 lone pairs, linear
B) 1 lone pair, bent
C) 2 lone pairs, bent
D) 3 lone pairs, bent
E) 3 lone pairs, linear
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
N,N-diethyl-m-tolumide (DEET) is the active ingredient in many mosquito repellents. What is the hybridization state of the nitrogen atom in the structure of DEET shown below 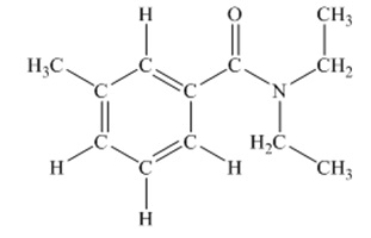
A) sp
B) sp2
C) sp3
D) sp3d
E) sp3d2
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the bond order of CO+
A) 1
B) 1.5
C) 2
D) 2.5
E) 3
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Indicate the type of hybrid orbitals used by the central atom in SF6.
A) sp
B) sp2
C) sp3
D) sp3d
E) sp3d2
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A sp2 hybridized central atom has what angles between its hybrid orbitals
A) 145
B) 120
C) 109.5
D) 90
E) < 90
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Showing 41 - 60 of 146
Related Exams