A) a charged atom or group of atoms with a net negative charge.
B) a stable atom.
C) a group of stable atoms.
D) an atom or group of atoms with a net positive charge.
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Essay
Name the following binary compound: AgCl.
Correct Answer

verified
silver chl...View Answer
Show Answer
Correct Answer
verified
View Answer
Short Answer
Write the formula for the acid formed from the permanganate anion, and then name the acid.
Correct Answer

verified
HMnO4, perm...View Answer
Show Answer
Correct Answer
verified
View Answer
Multiple Choice
Which of the following elements is chemically similar to oxygen?
A) sulfur
B) calcium
C) iron
D) nickel
E) sodium
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Short Answer
A molecule of antifreeze, ethylene glycol, has the formula C2H4(OH)2. How many atoms are there in one molecule of antifreeze?
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the formula for the ionic compound formed by calcium ions and nitrate ions?
A) Ca3N2
B) Ca(NO3) 2
C) Ca2NO3
D) Ca2NO2
E) CaNO3
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Short Answer
How many electrons, protons, and neutrons does an iron-55 atom have?
Correct Answer

verified
26 electro...View Answer
Show Answer
Correct Answer
verified
View Answer
Short Answer
What are the three types of radiation produced by the decay of substances like uranium?
Correct Answer

verified
Alpha, bet...View Answer
Show Answer
Correct Answer
verified
View Answer
Short Answer
Give the formula of copper(II)bromide.
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The Stock system name for CrO3 is:
A) chromium oxide
B) chromium(II) oxide
C) chromium(III) trioxide
D) chromium(III) oxide
E) chromium(VI) oxide
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Essay
The table below describes four atoms.  Which atoms represent the same element?
Which atoms represent the same element?
Correct Answer

verified
Atoms A and D repres...View Answer
Show Answer
Correct Answer
verified
View Answer
Short Answer
How many electrons, protons, and neutrons are in a neutral atom of the following isotope of krypton? 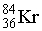
Correct Answer

verified
36 electro...View Answer
Show Answer
Correct Answer
verified
View Answer
Short Answer
Write the formula for the acid formed from the fluoride anion, and then name the acid.
Correct Answer

verified
HF, hydrof...View Answer
Show Answer
Correct Answer
verified
View Answer
Short Answer
Use the periodic table above to show where the metals are located.
Correct Answer

verified
Correct Answer
verified
Short Answer
Write the formula for the acid formed from the nitrite anion, and then name the acid.
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Consistent with vanadium being a transition metal, the name for VSO4 should be
A) vanadium sulfide.
B) vanadium (I) sulfite.
C) vanadium (I) sulfate.
D) vanadium (II) sulfate.
E) vanadium sulfur tetraoxide.
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The formula for magnesium sulfate is
A) MnS.
B) MgS.
C) MnSO3.
D) MgSO4.
F) A) and C)
Correct Answer

verified
Correct Answer
verified
True/False
When a beam of alpha particles passes between two electrically charged plates, the beam is deflected toward the positive plate.
B) False
Correct Answer

verified
Correct Answer
verified
Short Answer
Name the following ternary compound: CuCO3.
Correct Answer

verified
Correct Answer
verified
Short Answer
Consider a neutral atom of the following isotope of sulfur: 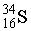 How many electrons, protons, and neutrons does the atom contain?
How many electrons, protons, and neutrons does the atom contain?
Correct Answer

verified
16 electro...View Answer
Show Answer
Correct Answer
verified
View Answer
Showing 41 - 60 of 141
Related Exams