A) none
B) one
C) two
D) three
E) four
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What orbitals overlap to create the H-C bond in CH3+?
A) sp3-sp3
B) sp2-sp3
C) s-p
D) s-sp2
E) s-sp3
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What type of bonding is most important in CH3CH2CH2CH2CH2CH3?
A) ionic
B) hydrogen
C) covalent
D) polar
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following structures, including formal charges, is correct for diazomethane, CH2N2?
A) : CH2-N ![]() N :
N :
B) ![]()
C) ![]()
D) ![]()
E) ![]()
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Short Answer
Covalent bonds may be polar or nonpolar. What property of the atoms forming a given bond determines this?
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The compound methylamine, CH3NH2, contains a C-N bond. In this bond, which of the following best describes the charge on the nitrogen atom?
A) +1
B) slightly positive
C) uncharged
D) slightly negative
E) -1
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following is the most likely electronic structure for C2H2?
A) ![]()
B) ![]()
C) ![]()
D) H-C ![]() C-H
C-H
E) ![]()
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Identify the compound with the weakest bond.
A) H2
B) HF
C) HCl
D) HBr
E) HI
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Identify the least electronegative atom.
A) P
B) Na
C) I
D) B
E) O
G) All of the above
Correct Answer

verified
Correct Answer
verified
Short Answer
Give the hybridization, shape, and bond angle for the carbon in CO2.
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following molecules has a net dipole moment of zero?
A) ![]()
B) ![]()
C) ![]()
D) ![]()
E) ![]()
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following molecules has the smallest dipole moment?
A) Br2
B) NH3
C) HCl
D) HBr
E) HI
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Essay
The Kekulé structure of pentane is shown below. Draw the condensed structural formula which corresponds to this Lewis structure. 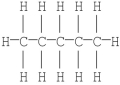
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following is the electronic configuration of the element Fe?
A) 1s2 2s2 2p6 3s2 3p6 4s2 3d6
B) 1s2 2s2 2p6 3s2 3p8 3d6
C) 1s2 2s2 2p8 3s2 3p6 4s2 3d6
D) 1s2 2s2 2p6 3s2 3p6 4s2 4d6
E) 1s2 2s2 2p6 3s2 3p6 4s2 4p6
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Essay
Draw the Kekulé structure for each of the following: a. CH3CH2OH b. CH3CHO c. (CH3)3C+
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following covalent bonds has the largest dipole moment?
A) C-C
B) C-H
C) C-O
D) H-N
E) H-F
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How many distinct and degenerate p orbitals exist in the second electron shell, where n = 2?
A) 0
B) 1
C) 2
D) 3
E) 4
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the compounds below have bonds that are predominantly ionic?
A) KCl
B) CF4
C) NH3
D) both A and B
E) both B and C
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Short Answer
Identify the hybridization of the nitrogen atom in the molecule (CH3)3N.
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The carbon-carbon double bond in ethene is ________ and ________ than the carbon-carbon triple bond in ethyne.
A) stronger; shorter
B) stronger; longer
C) weaker; shorter
D) weaker; longer
E) stronger; more polar
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Showing 61 - 80 of 81
Related Exams